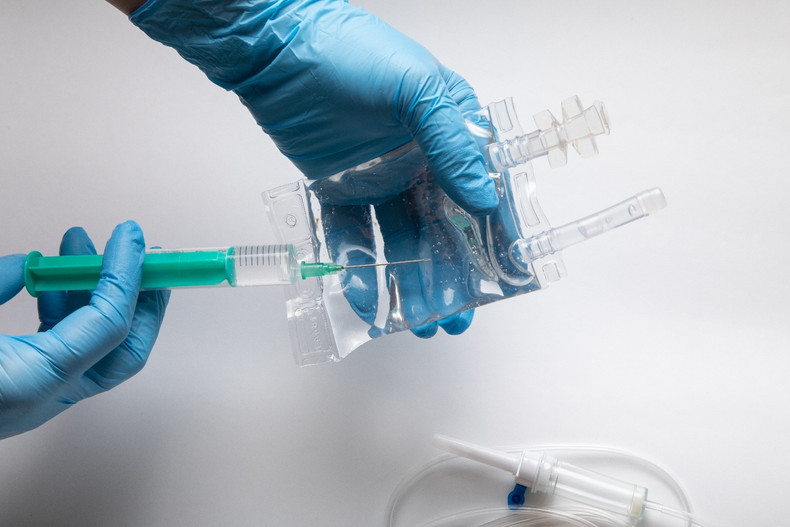
Intravenous (IV) infusion therapy is one of the most common and critical procedures in healthcare settings. It is used for administering fluids, medications, nutrients, and blood products directly into a patient’s bloodstream. Because it bypasses the body’s natural protective barriers, IV therapy demands a high level of precision, hygiene, and consistency. Even minor lapses in quality can lead to serious complications such as infections, dosage errors, or contamination.
For hospitals, clinics, and care facilities, maintaining safe IV infusion practices is not only a clinical responsibility but also a supply chain concern. The role of medical consumable suppliers becomes especially important in ensuring that every component used in IV therapy meets strict quality standards.
The Importance of Quality in IV Infusion
IV infusion involves multiple consumable components such as catheters, tubing sets, cannulas, infusion bags, connectors, and syringes. Each item must meet safety requirements because they all come into direct or indirect contact with the patient’s bloodstream.
Unlike some medical procedures where equipment failure might be inconvenient, IV-related issues can escalate quickly into life-threatening situations. This is why healthcare providers rely heavily on consistent sourcing from trusted medical consumable suppliers who understand the critical nature of these products.
Quality in IV infusion is not just about product durability—it is about sterility, compatibility, and predictable performance under clinical conditions.
Sterility and Infection Control Standards
One of the most important quality standards in IV infusion is sterility. Every consumable used in the process must be free from microorganisms, pyrogens, and contaminants.
Single-use packaging is commonly used to ensure sterility is maintained until the point of use. Proper sealing, validated sterilization processes, and tamper-evident packaging all contribute to infection prevention.
Healthcare facilities also follow strict protocols during handling and administration, but even the most careful practices depend on the integrity of products supplied. This is where dependable medical consumable suppliers play a key role by ensuring that sterilization processes meet international benchmarks and are consistently applied across batches.
Material Safety and Biocompatibility
IV infusion materials must be biocompatible, meaning they should not cause adverse reactions when in contact with human tissue or blood. This includes avoiding toxic substances such as certain plasticizers or latex components that may trigger allergies.
Modern IV consumables are typically manufactured using medical-grade PVC, polyurethane, or silicone-based materials that have been tested for safety.
A reliable supply chain ensures that materials remain consistent across production runs. Variability in material quality can lead to unpredictable patient responses, which is why procurement teams must carefully evaluate the sourcing practices of medical consumable suppliers before making long-term agreements.
Precision Engineering and Flow Control
IV therapy often requires precise control of fluid flow rates, especially when administering medications with narrow therapeutic ranges. Infusion sets must therefore be designed to deliver accurate and stable flow without leakage or blockage.
Components like drip chambers, flow regulators, and connectors must meet engineering standards that support accuracy under different pressure conditions.
Poorly manufactured consumables can lead to underdosing or overdosing, both of which pose serious risks to patient safety. This makes product consistency a key factor when selecting suppliers and manufacturers in the healthcare procurement process.
Regulatory Compliance and Certification
Quality standards in IV infusion are heavily regulated by health authorities and international organizations. Certifications such as ISO standards for medical devices, CE marking in certain regions, and compliance with Good Manufacturing Practices (GMP) are essential indicators of product reliability.
Healthcare providers must ensure that every product used in IV therapy is backed by proper documentation and traceability. This includes batch numbers, expiration dates, and sterilization records.
Trusted medical consumable suppliers typically maintain transparent compliance systems that allow healthcare institutions to verify product authenticity and regulatory approval before use.
Storage, Handling, and Shelf Life Management
Even high-quality IV consumables can become unsafe if not stored or handled correctly. Temperature, humidity, and exposure to contaminants can all affect product integrity.
Suppliers must therefore provide clear storage guidelines and ensure that packaging protects products throughout the supply chain. Healthcare facilities, on their part, must manage inventory effectively to prevent the use of expired or compromised materials.
Shelf life tracking is particularly important in IV therapy consumables, where expired products can lose sterility or structural integrity.
Role of Reliable Supply Chains in Patient Safety
Behind every safe IV infusion procedure is a well-functioning supply chain. Disruptions in availability or inconsistencies in product quality can directly affect patient care.
This is why healthcare institutions prioritize long-term partnerships with dependable medical consumable suppliers who can guarantee continuity, quality assurance, and timely delivery.
A strong supply chain also helps hospitals respond to emergencies, manage high patient volumes, and maintain uninterrupted clinical operations.
Training and Standardization Support
Beyond supplying products, many leading suppliers contribute to healthcare safety by offering training resources and technical guidance. This helps clinical staff understand correct usage techniques, reduce procedural errors, and stay updated with evolving standards.
Standardization across consumables also simplifies training and reduces variability in clinical practice, improving overall safety outcomes.
Conclusion
Safe IV infusion practices depend on a combination of skilled healthcare professionals, strict clinical protocols, and, most importantly, high-quality consumables. From sterility and material safety to precision engineering and regulatory compliance, every detail matters.
Selecting trustworthy medical consumable suppliers is not just a procurement decision—it is a patient safety decision. When quality is consistently prioritized across the supply chain, healthcare providers can significantly reduce risks and deliver more reliable, effective care to every patient.